Immune response and endothelial dysfunction
Dicken Weatherby, N.D. and Beth Ellen DiLuglio, MS, RDN, LDN
Immune response and infiltration of immune cells can promote endothelial dysfunction and atherosclerosis.
The Endothelial Dysfunction Series
- Endothelial Dysfunction part 1 - An Overview
- Endothelial Dysfunction part 2 - The Endothelium
- Endothelial Dysfunction part 3 - Nitric Oxide
- Endothelial Dysfunction part 4 - Diseases and Causes
- Endothelial Dysfunction part 5 - Immune Response & Oxidative Stress
- Endothelial Dysfunction part 6 - Atherosclerosis
- Endothelial Dysfunction part 7 - Assessment Part 1
- Endothelial Dysfunction part 8 - Assessment part 2
- Endothelial Dysfunction part 9 - Functional Naturopathic Approach
- Endothelial Dysfunction part 10 - Optimal Takeaways
Neutrophils are the most abundant white blood cells of the immune system.[i] These multi-purpose cells can be both protective and destructive. On the downside, their contribution to atherosclerosis has been overlooked. They exude neutrophil extracellular traps (NETs) that trap pathogens but can also trigger endothelial dysfunction, inflammation, and atherosclerosis. NETs have been identified in atherosclerotic plaque.
Researchers suggest that neutrophil counts are significant predictors of cardiovascular events.[ii]
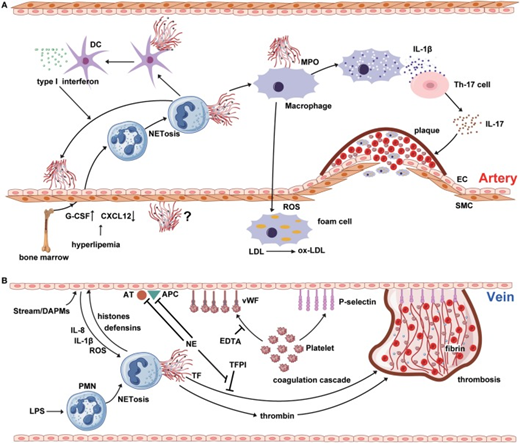
NETosis interweaves atherosclerosis and thrombosis.
- Neutrophil extracellular traps (NETs) are involved in the whole process of atherosclerosis. The myeloperoxidase from NTEs can stimulate macrophage to oxidize low-density lipoprotein (LDL) to ox-LDL and form the foam cell. The hyperlipidemia recruits neutrophil into circulation from bone marrow by upregulating the expression of granulocyte colony-stimulating factor and downregulating the level of C-X-C motif ligand -12, which is an important signal for the clearance and recruitment of aged neutrophils to the bone marrow.
- Cholesterol crystals can trigger the polymorphonuclear neutrophil (PMN) to release the NETs that prime the macrophages for pro-inflammatory cytokine production including IL-1β. Then IL-1β activates Th17 cell to release interleukin-17, amplifying the immune cell recruitment into the atherosclerotic plaque.
- As another critical source of foam cell, SMC also takes part in atherosclerosis. However, there are few reports about the interaction between NETs and SMC.
- (B) NETs are released from PMNs, which are activated by LPS or other cytokines from injured endothelial cells.
Source: Qi, Haozhe et al. “Neutrophil Extracellular Traps and Endothelial Dysfunction in Atherosclerosis and Thrombosis.” Frontiers in immunology vol. 8 928. 7 Aug. 2017, doi:10.3389/fimmu.2017.00928 [R] This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
Oxidative stress contributes to endothelial dysfunction due to:[iii] [iv]
- Increased reactive oxygen species (ROS) reduce NO bioavailability
- Increased oxidized LDL cholesterol (Ox-LDL)
- Reduced superoxide dismutase (SOD) (SOD can clear ROS)
- Oxidative stress can occur from an excess of oxidative factors and/or an insufficiency of antioxidants
- Intracellular ROS and oxidative stress contribute to the chronic inflammation seen in atherosclerosis[v]
- Hyperlipidemia, hyperglycemia, and hypertension can increase ROS
Oxidative stress and inflammation negatively affect nitric oxide metabolism, as do other factors that disrupt NO and lead to “global vasoconstriction.” These factors include mental stress, anger, and cold temperatures.[vi]
Reactive oxygen species (ROS) associated with oxidative stress can combine with nitric oxide to produce highly reactive molecules that in turn generate more oxidative stress, more atherosclerosis, and more vascular injury.[vii] [viii]
Oxidative stress can also reduce availability of nitric oxide by promoting excess degradation of tetrahydrobiopterin. Tetrahydrobiopterin (BH4) is a cofactor for crucial metabolic enzymes including nitric oxide synthase (NOS). It is therefore essential to the maintenance of endothelial function and a healthy blood pressure.
Tetrahydrobiopterin also plays a role in monoamine neurotransmitter synthesis, sensitivity to pain, and immune function. Research suggests that BH4 may be instrumental in restoring NO-redox balance and improving hyperlipidemia, ischemia-reperfusion injury, hypertension, and cardiac hypertrophy.[ix] [x] [xi]
Administration of BH4 and arginine in subjects with coronary artery disease (CAD) and impaired glucose tolerance/diabetes significantly improved endothelium-dependent vasodilation following ischemia reperfusion.[xii]
However, supplemental BH4 may be oxidized to dihydrobiopterin (BH2), posing a challenge to its efficacy.[xiii]
NEXT UP - Endothelial Dysfunction part 6 - Atherosclerosis
Research
[i] Rosales, Carlos. “Neutrophil: A Cell with Many Roles in Inflammation or Several Cell Types?.” Frontiers in physiology vol. 9 113. 20 Feb. 2018, doi:10.3389/fphys.2018.00113 [R]
[ii] Qi, Haozhe et al. “Neutrophil Extracellular Traps and Endothelial Dysfunction in Atherosclerosis and Thrombosis.” Frontiers in immunology vol. 8 928. 7 Aug. 2017, doi:10.3389/fimmu.2017.00928 [R] This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
[iii] Sena, Cristina M et al. “Vascular Oxidative Stress: Impact and Therapeutic Approaches.” Frontiers in physiology vol. 9 1668. 4 Dec. 2018, doi:10.3389/fphys.2018.01668 [R]
[iv] Leopold, Jane A, and Joseph Loscalzo. “Oxidative mechanisms and atherothrombotic cardiovascular disease.” Drug discovery today. Therapeutic strategies vol. 5,1 (2008): 5-13. doi:10.1016/j.ddstr.2008.02.001 [R]
[v] Yang, Rui-Li et al. “Increasing Oxidative Stress with Progressive Hyperlipidemia in Human: Relation between Malondialdehyde and Atherogenic Index.” Journal of clinical biochemistry and nutrition vol. 43,3 (2008): 154-8. doi:10.3164/jcbn.2008044 [R]
[vi] Widmer, R Jay, and Amir Lerman. “Endothelial dysfunction and cardiovascular disease.” Global cardiology science & practice vol. 2014,3 291-308. 16 Oct. 2014, doi:10.5339/gcsp.2014.43 [R] This is an open access article distributed under the terms of the Creative Commons Attribution license CC BY 4.0, which permits unrestricted use, distribution and reproduction in any medium, provided the original work is properly cited.
[vii] Kattoor, Ajoe John et al. “Oxidative Stress in Atherosclerosis.” Current atherosclerosis reports vol. 19,11 42. 18 Sep. 2017, doi:10.1007/s11883-017-0678-6 [R]
[viii] Salvayre, R et al. “Oxidative theory of atherosclerosis and antioxidants.” Biochimie vol. 125 (2016): 281-96. doi:10.1016/j.biochi.2015.12.014 [R]
[ix] Moens, An L, and David A Kass. “Tetrahydrobiopterin and cardiovascular disease.” Arteriosclerosis, thrombosis, and vascular biology vol. 26,11 (2006): 2439-44. doi:10.1161/01.ATV.0000243924.00970.cb [R]
[x] Bendall, Jennifer K et al. “Tetrahydrobiopterin in cardiovascular health and disease.” Antioxidants & redox signaling vol. 20,18 (2014): 3040-77. doi:10.1089/ars.2013.5566 [R]
[xi] Zhao, Yuwen et al. “Detection of tetrahydrobiopterin by LC-MS/MS in plasma from multiple species.” Bioanalysis vol. 1,5 (2009): 895-903. doi:10.4155/bio.09.77 [R]
[xii] Settergren, M et al. “L-arginine and tetrahydrobiopterin protects against ischemia/reperfusion-induced endothelial dysfunction in patients with type 2 diabetes mellitus and coronary artery disease.” Atherosclerosis vol. 204,1 (2009): 73-8. doi:10.1016/j.atherosclerosis.2008.08.034 [R]
[xiii] Yuyun, Matthew F et al. “Endothelial dysfunction, endothelial nitric oxide bioavailability, tetrahydrobiopterin, and 5-methyltetrahydrofolate in cardiovascular disease. Where are we with therapy?.” Microvascular research vol. 119 (2018): 7-12. doi:10.1016/j.mvr.2018.03.012 [R]






